In many parts of the world, legislation has been and is being enacted to counter the increasing number of counterfeit medicines. To make counterfeit products more difficult and to increase patient safety, pharmaceutical manufacturers will have to provide all affected drugs with a globally unique serial number code.
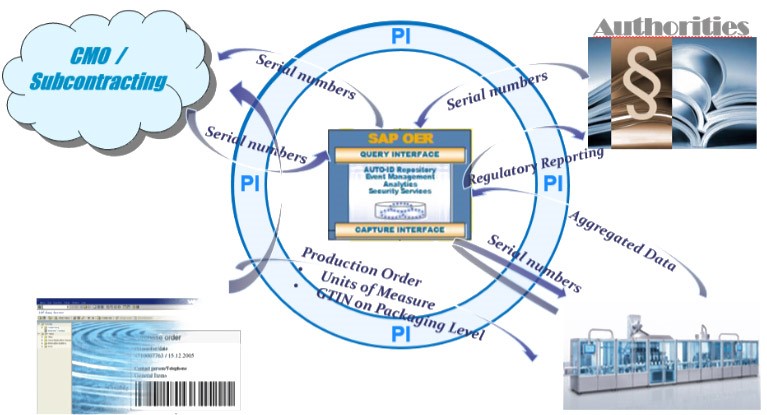
As a result, each package of a prescription medicine must be provided with a unique and unambiguous identifier (serial number). In addition, the serialisation data for each individual package must be assigned, recorded and made available in accordance with the länderspezifischen regulations of the target market. Effective, transparent and centralized data management is required to verify and track serialized products throughout the manufacturing and supply chain. This includes all systems, manufacturing, packaging and logistics processes up to the Enterprise Resource Planning (ERP) systems, and essentially distinguishes between two approaches:
- In the end-to-end approach, the serial number is only applied to the sales unit
- With the Track & Trace approach, every change of ownership along the supply chain must be registered. Thus, in addition to the sales unit, each outer packaging must also be registered with an
serial number can be provided, recorded and saved
The SAP OER (Object Event Repository) offers a suitable solution in the environment of an SAP system landscape, which is to be regarded as the core of a serialization solution. It assigns and manages all serial numbers and stores all their status changes.
We offer you our experience (especially in the pharmaceutical industry) and our know-how in all modules regarding serialization:
Project Management
- Technical Project/Process Planning
- Coordination/coordination with your production line manufacturers
- Country-Specific Reporting
- Number Range and List Management
- Necessary adjustments in the SAP modules
- Object Event Repository (OER)
- Material Management (MM)
- Process planning (PP)
- Sales (SD)
- Warehouse management (WM)
Customizing and development
- OER
- AII

